Denmark’s trial scheme for medical cannabis will be extended beyond the end of 2021, when the temporary program had been set to expire, the country’s Ministry of Health announced Tuesday.
The trial gives doctors the ability to prescribe cannabis to patients and appropriately licensed businesses the ability to grow and ship medical marijuana.
A majority of parliamentary parties agreed to make permanent the ability for businesses to grow cannabis for medical use – a welcome move for companies such as Alberta-based Aurora Cannabis, which still operates a production subsidiary in Denmark.
“The permanent authorization for licensed producers of the production and the export of medical cannabis in Denmark, independent of the pilot program, is an important milestone recognizing this young industry,” the Danish subsidiary of Aurora said in a statement to MJBizDaily.
Other Canadian companies, however, are planning to leave the country or have already reduced their presence there.
The parliamentary agreement also means doctors will continue to have the ability to prescribe cannabis to patients for another four years.
Bills will be adopted before the end of this year to implement the agreements, according to the ministry’s statement.
The proposed changes come after Denmark’s medical cannabis program has faced significant challenges in recent years, underscoring why cannabis producers need to factor ebbs and flows into their market projections rather than counting on perpetual growth.
Early success fades
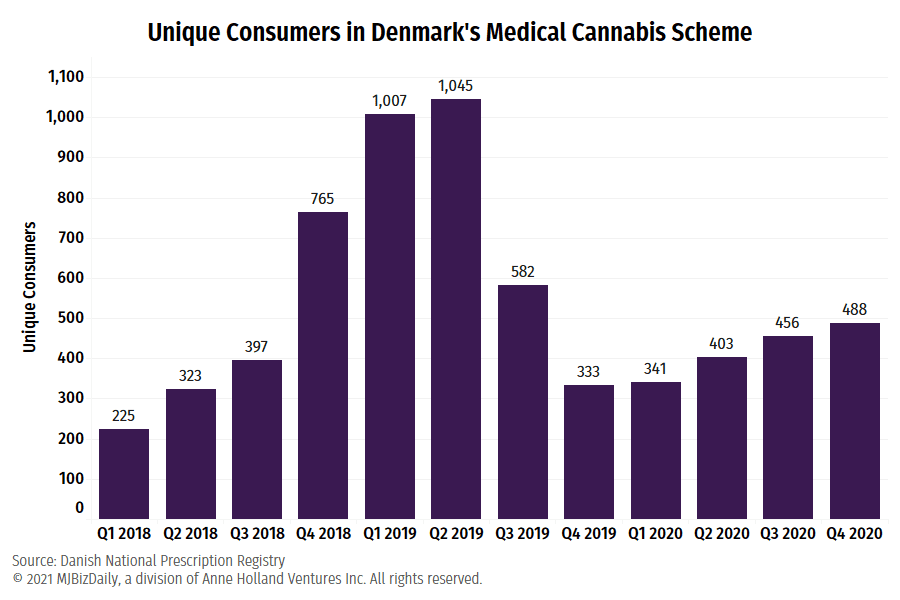
The pilot enjoyed rapid growth in patient uptake in its first two years.
A little more than 1,700 patients were approved for medical cannabis in 2018, the program’s first year. That rose to 2,967 patients the following year, before falling to fewer than 1,700 patient approvals for 2020.
At the program’s peak in mid-2019, over 1,000 patients received 1,800 prescriptions in consecutive quarters.
In the final three months of last year, however, fewer than 500 patients accessed medical cannabis through the trial.
Getting products permitted for local sale has been difficult.
Only eight of the 63 applications to admit products to the trial scheme had won approval by mid-2020, MJBizDaily reported last year.
All medical cannabis provided to Danish patients was imported.
Access to cannabis produced within Denmark has been another hurdle.
Still no locally made products
Earlier this year, a subsidiary of Canada’s Aurora Cannabis said its “made in Denmark” medical cannabis products reached Germany pharmacies for the first time – a milestone that was years in the making.
But that came before any cannabis produced in Denmark had reached local patients, even though the trial had been up-and-running for more than three years.
Only four products are available in Denmark under the experiment, including one of Aurora’s.
Despite the challenges, Aurora’s Danish subsidiary said in its statement the company is “very pleased” the Danish pilot program will be extended.
The statement also noted that in January 2018, Aurora was one of the first companies to invest in Denmark – initially as a joint venture and today as a fully owned subsidiary of Aurora Cannabis.
Marianne Hundtofte Nielsen, vice president of business development for Aurora Europe, said the company currently sells Sedemen capsules in Denmark that are imported from Canada.
“Aurora Nordic is working closely with the local regulatory authority (Danish Medicines Agency) to offer a number of treatment options, such as extracts and dried flowers, to Danish physicians and patients, as part of the cannabis pilot program,” she said in a statement.
In a regulatory filing, the company had said it expected products made in Denmark to be available for sale in mid-2019.
Canadians scale back
Facing rising capital expenditures in Denmark, several Canadian companies have scaled back ambitious expansion plans there.
In late 2019, Aurora ceased construction at its large second-phase facility in Denmark to save money.
Canadian cannabis producer Canopy Growth is in the process of selling its Danish facility.
“Canopy Growth remains in the consultation period regarding the sale of its operations in Denmark,” a spokesperson told MJBizDaily via email.
Canopy said it will continue to supply the European market through Canadian exports.
The sale strictly involves the facility.
“Upon closure of this transaction, the facility will continue to operate, and the new operator has expressed their desire to retain all 60 of the current employees.”
Shares of Aurora trade as ACB on the Nasdaq and Toronto Stock Exchange.
Matt Lamers is Marijuana Business Daily’s international editor, based near Toronto. He can be reached at mattl@mjbizdaily.com.